Researcher uses funding to study heavy metal tolerance
By Sarah Perdue
Why is it that some people develop cancer, Alzheimer's or Parkinson's disease after chronic exposure to high amounts of heavy metals, but other people tolerate exposure without developing these diseases?
Using the worm model system C. elegans and a grant of almost $750,000 from the National Science Foundation funded by the American Reinvestment and Recovery Act (ARRA), Olena Vatamaniuk, assistant professor in the Department of Crop and Soil Sciences, hopes to answer that question.
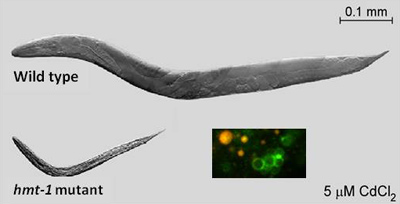
"We are trying to understand how heavy metals and byproducts of heavy metal toxicity are detoxified," said Vatamaniuk. Almost all species, including humans and C. elegans, have several genes that allow for the detoxification of heavy metals. Vatamaniuk focuses on one gene in particular, HMT-1 (heavy metal tolerance factor 1). Worms that lack HMT-1 cannot survive when exposed to cadmium, but worms that have the gene do survive.

"The question is how this protein detoxifies during heavy metal exposure," she said.
Heavy metals themselves are not directly toxic, but they can damage DNA, proteins and other cellular molecules. Vatamaniuk's previous work has shown that HMT-1 functions by pumping some unknown molecules into cellular compartments where they are sequestered. The new funding will help her elucidate more specifically how HMT-1 prevents damage from heavy metals by identifying which substances are targeted for sequestration.
HMT-1 belongs to a family of proteins called ABC transporters; they get into the membrane of cells and can transport molecules in and out of the cell. While some of these proteins do not need other ABC transporters to function, HMT-1 (among others) does.
One of the main focuses of the grant is to identify which other ABC transporter proteins -- as well as proteins that are not ABC transporters -- HMT-1 works with to prevent heavy metal toxicity.
"You can relate the function of the interacting proteins to the function of HMT-1 and learn how HMT-1 can contribute to detoxification," Vatamaniuk said. "This work will also allow us to study the role of other ABC transporters in heavy metal detoxification as compared to HMT-1."
Vatamaniuk will use study C. elegans to study HMT-1 because it is a relatively simple organism and because of the tissues that express this protein.
"HMT-1 in C. elegans is expressed in all cell types that are affected the most by heavy metal poisoning in humans, including neurons and intestinal cells, and also unique cells for C. elegans that function as a primitive liver," she said. "If we can understand the function of HMT-1 using a simple organism like C. elegans, then we can relate this function to higher organisms like us."
With the stimulus grant, Vatamaniuk plans to hire two postdoctoral fellows and train graduate and undergraduate students. She will also purchase such state-of-the-art equipment as fluorescent microscopes to further the research.
Because such farming practices as the application of fertilizer and pesticides can lead to heavy metal accumulation in soil, part of the funding will also go toward educational outreach. Vatamaniuk and her research group will collaborate with the Cornell Nutrient Analysis Lab and the Cornell Waste Management Institute to reach New York farmers and gardeners to discuss the importance of testing soils for contaminants. "Heavy metals are not biodegradable, and once they are accumulated they can be incorporated into the food chain," she said, adding that most soil testing in upstate New York does not measure heavy metal contamination.
To date, Cornell has received 109 ARRA awards totaling $93 million.
Graduate student Sarah Perdue is a science writer intern for the Cornell Chronicle.
Get Cornell news delivered right to your inbox.
Subscribe