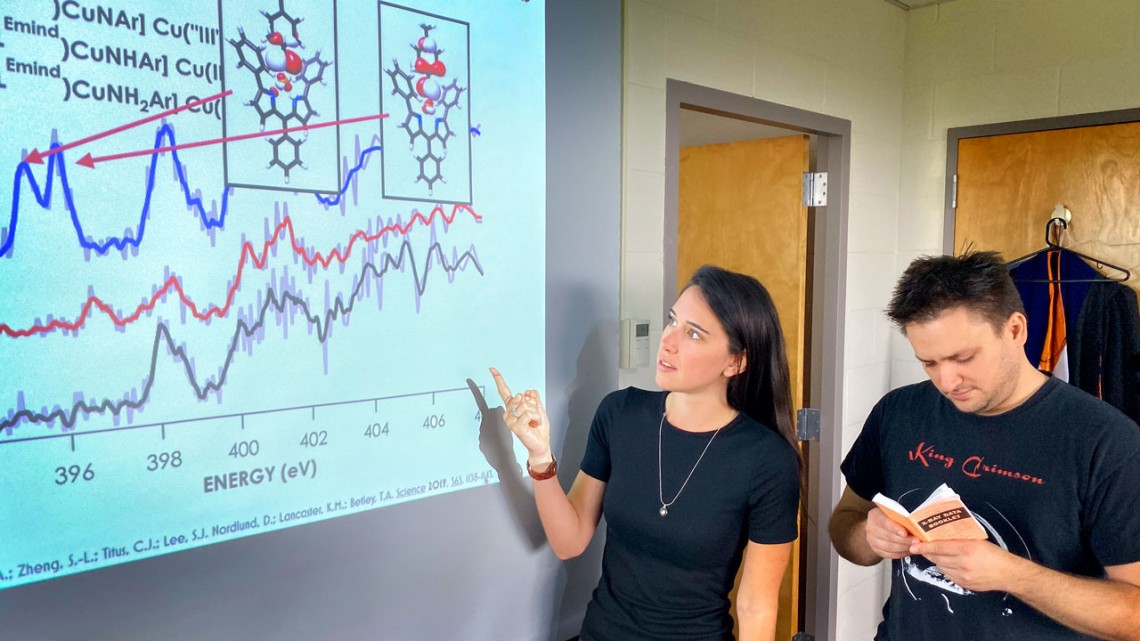
Doctoral student Ida DiMucci and Kyle Lancaster, associate professor of chemistry and chemical biology, review the spectroscopic data supporting their identification of a copper-nitrene catalyst.
Missing electrons reveal the true face of a new copper-based catalyst
By David Nutt
Some chemical bonds cling more tightly than others. Chummy carbon-hydrogen bonds, for example, are notoriously difficult to separate.
A collaboration between researchers from Cornell, Harvard, Stanford and the SLAC National Accelerator Laboratory has resulted in a reactive copper-nitrene catalyst that pries apart carbon-hydrogen (C–H) bonds and transforms them into carbon-nitrogen (C–N) bonds, which are a crucial building block for chemical synthesis, especially in pharmaceutical manufacturing.
The team’s paper, “Synthesis of a Copper–Supported Triplet Nitrene Complex Pertinent to Copper–Catalyzed Amination,” was published Sept. 13 in Science.
“[Co-senior author] Ted Betley at Harvard and I are both interested in cases where you have light atoms like nitrogen or oxygen that typically are well defined in the oxidation state,” said co-senior author Kyle Lancaster, associate professor of chemistry and chemical biology in the College of Arts and Sciences, who worked on the project with his Ph.D. student Ida DiMucci.
“We found that when you oxidize this one copper compound that has this nitrogen functionality to it, you pull the electrons out of the nitrogen. When you have light atoms like nitrogen or oxygen that are electron deficient, that tends to make them very reactive,” Lancaster said. “And in this case, the nitrogen is so reactive it is able to break C–H bonds and start making C–N bonds.”
While researchers have long suspected that this type of nitrogen that lacks two specific electrons – a nitrene – was tied to reactivity, the elusive species had yet to be directly spotted. Among the specialties of Lancaster’s lab is employing X-ray spectroscopy to determine where electrons are missing in transition metal-containing compounds. Lancaster and DiMucci went to work inventorying the electrons on the copper and the nitrogen atoms and were able to clear away some of the atomic clutter and zero in on the nitrogen to locate two holes that indicated where the two electrons were being lost.
Lancaster and DiMucci performed some high-level computational analysis to confirm their findings.
“We were really stunned how strong the agreement was between the theory we used and our data,” DiMucci said. “Our calculations told us that if we really had what we thought we did, then we’d be looking for two peaks separated by 0.6 electron volts, and that’s exactly what we got.”
The Harvard team constructed the ligand structure to secure the reactive nitrene and keep the catalyst together. Once a C–H bond is introduced, the nitrene is able to unshackle it. This streamlined conversion process could lead to cheaper, more efficient production of pharmaceuticals, detergents and dyes.
“At the end of the day, when we find a big, hairy organic molecule, we’d like to be able to make it en masse if it’s a potential drug,” Lancaster said. “And that means coming up with clever ways to install particular functionalities, like C–O bonds and C–N bonds. The more tools we have in our arsenal to make these changes – because it’s very difficult to turn the C–H bond into a C–N bond – the better off we are.”
Lancaster praised the work of his Harvard, Stanford and SLAC colleagues, and the potential applications of their collaborative work.
“We all have our specializations,” he said, “and teaming up spectroscopy and electronic structure theory with synthesis is a powerful combo to answer these chemical questions – especially when you have physicists coming up with exquisite new instruments to make our job a lot easier.”
The Cornell research was supported by National Science Foundation and the Alfred P. Sloan Foundation.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe
