3-D printed artificial gut creates realistic model for research
By Krishna Ramanujan
One challenge for studying intestinal biology is there hasn’t been a realistic model that re-creates the conditions in a living intestine.
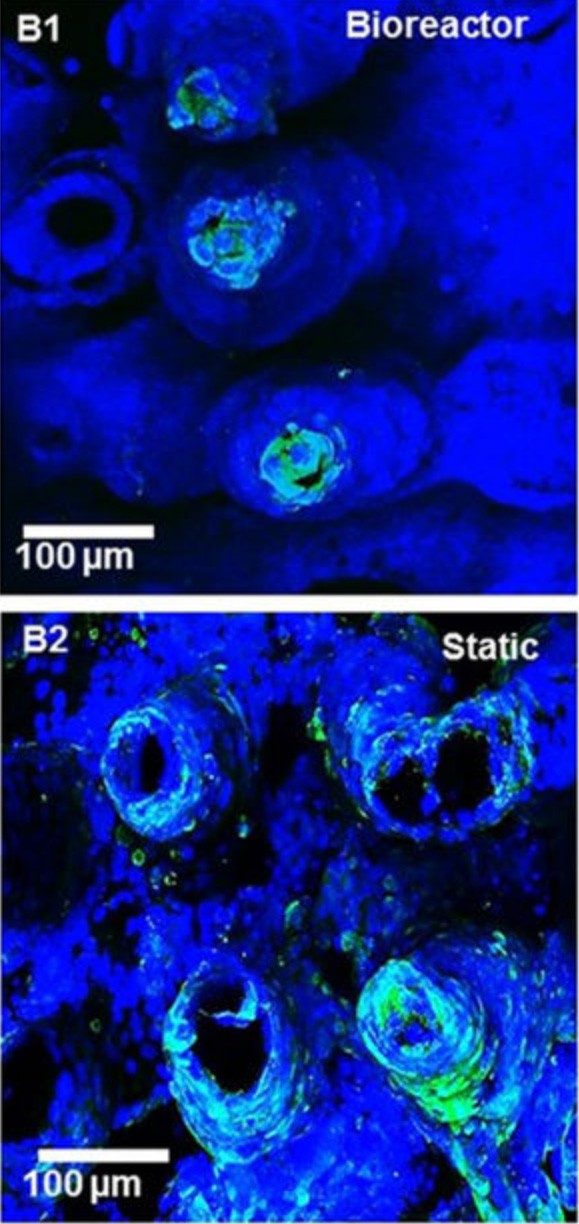
Cornell researchers have used 3-D printing technology to create a microscale artificial small intestine that realistically mimics the gut surface topography (texture and structure) and fluid flow that epithelial (gut surface) cells require to grow, reproduce and function properly. The artificial intestine re-creates peristaltic fluid flow, which other models have failed to achieve.
Since the artificial intestine is 3-D printed, almost any research group can now make one and tailor such systems to its specific needs.
“The intestine is home to most of our immune system and it is also where all of our nutrients are absorbed, so to function as a human being or any multicellular animal, our gut is the centerpiece of our interactions with the outside world,” said John March, professor and chair of the Department of Biological and Environmental Engineering. “The more we understand about how our guts work, the more we can develop therapies and gain insights into the evolution of human biology.”
March is the senior author of a paper describing this system, published Oct. 2 in Scientific Reports. Cait Costello, a research associate in March’s lab, is the study’s first author.
The new 3-D printed system allows researchers to understand the dynamics of fluid flow in the intestine. The study showed that recreating peristaltic flow in the model allowed cells to grow as they would in a living intestine. Proper flow encourages cells to die off selectively along the tips of intestinal villi. When such flow is missing, cells all along villi become self-destroying. Also, accurate fluid flow lets researchers study dynamics of bacterial populations in the intestines, as some bacteria are better able to deal with flow than others.
Co-authors include researchers at Johns Hopkins University, the National University of Singapore and the University of Maryland.
The study was funded by the National Institutes of Health.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe