Researchers chart the contents of human bone marrow
By Wynne Parry
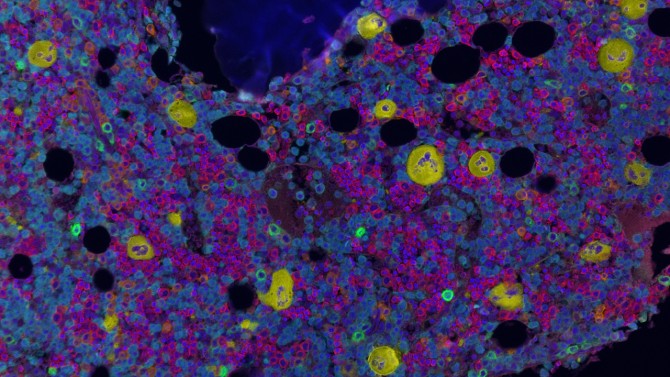
A team at Weill Cornell Medicine has mapped the location and spatial features of blood-forming cells within human bone marrow. Their findings confirm hypotheses about the anatomy of this tissue and provide a powerful new means to study diseases that affect bone marrow, ranging from noncancerous conditions such as sickle cell anemia to malignant conditions such as acute leukemia.
For the research described Sept. 29 in Blood, the investigators retrieved deidentified archival bone marrow samples from 29 patients at NewYork-Presbyterian/Weill Cornell Medical Center, generating a vast amount of data about the spatial relationships among their contents.
Creating images of bone marrow has been difficult historically, according to senior author Dr. Sanjay Patel, director of the Multiparametric In Situ Imaging (MISI) Laboratory in the Department of Pathology and Laboratory Medicine and an assistant professor of pathology and laboratory medicine at Weill Cornell Medicine. He and his colleagues overcame these challenges by devising a method for visualizing whole pieces of the tissue, then analyzing them with artificial intelligence (AI).
“We have been able to apply our approach to archival samples in a way that wasn’t possible before,” said Patel, who is also a hematopathologist at NewYork-Presbyterian/Weill Cornell Medical Center and a member of the Sandra and Edward Meyer Cancer Center at Weill Cornell Medicine. He noted that they succeeded in identifying and determining the positions of about 1.5 million cells in all.
Our blood cells get their start in the bone marrow, where stem cells produce the progenitors that in turn generate red and white blood cells, as well as the wound-sealing fragments known as platelets. Errors in these processes can give rise to acquired diseases including cancers, such as leukemia, lymphoma and multiple myeloma, and those, such as sickle cell anemia, present from birth.
Studying the birth of blood cells within their native environment in human tissues, however, has proven challenging. What’s more, when bone marrow samples are collected, the preservation technique can degrade some nucleic acids and proteins within the cells they contain. And, to avoid bias, researchers need to capture images of an entire piece of tissue, generating a daunting amount of data.
Patel’s team came up with a series of solutions. The researchers started by gathering samples from the tissue archive within Weill Cornell Medicine’s Department of Pathology and Laboratory Medicine. These 1- to 2-centimeter pieces of tissue came from patients who had received biopsies, but who had turned out to be disease free. Researchers in the MISI lab tested a variety of immune proteins known as antibodies, selecting from a catalog of thoroughly vetted markers used in routine clinical diagnostics, to see which most effectively tagged the contents of bone marrow to make them visible with their fluorescence-based imaging instrumentation.
Their collaborators at BostonGene Corporation, a medical bioinformatics company, then used AI to analyze the resulting images, picking out individual cells, such as stem cells and the platelet-producing megakaryocytes, as well as bone, fat and blood vessels. This technology allowed the team to wrangle an otherwise unmanageable amount of information into a sophisticated analysis, according to Patel.
Previous studies have suggested that, during normal blood cell development, stem and progenitor cells inhabit certain locations, near bone and blood vessels, where surrounding cells create environments critical for their normal function. More recently, some research has suggested that these cells also gather around megakaryocytes, large cells that give rise to platelets. The team’s analysis confirmed these patterns, including for megakaryocytes, in human samples. However, when they took patients’ age into account, they found the cells were no longer as closely associated with megakaryocytes, which also tended to be smaller in older patients.
While these findings contribute to scientists’ understanding of normal bone marrow, Dr. Patel sees the new method’s greatest potential in investigating diseases, particularly along the course of their evolution. For a few conditions, such as acute myeloid leukemia, researchers already have evidence that the spatial arrangement of stem and progenitor cells may be disrupted. This new method could open the door to studies that specifically explore such changes – and to those testing new treatments and evaluating existing ones, according to Patel.
“I hope our work unlocks the imagination of people who study diseases related to the bone marrow,” he said.
Wynne Parry is a freelance writer for Weill Cornell Medicine.
Media Contact
Eliza Powell
Get Cornell news delivered right to your inbox.
Subscribe