Window over mouse spinal cord allows imaging to aid trauma therapy
By Krishna Ramanujan
To study spinal cord injuries, researchers have had to conduct exploratory surgeries on mice to determine how nerves and other cells respond after trauma. But these approaches have only shown snapshots in time and have failed to provide key, game-changing information.

But now, Cornell researchers have developed an imaging technique similar to one used to study the brain, in which a small portion of a mouse skull is replaced with glass so scientists can observe the brain to study Alzheimer's disease, strokes and brain cancers.
The new spinal cord procedure, described in the Jan. 22 online issue of the journal Nature Methods, involves surgically implanting a window, or chamber with a transparent panel over a live mouse's exposed cord. Fluorescently labeled objects, such as neural cells or blood vessels, are then visualized in 3-D using multiphoton microscopy (invented by Cornell biophysicist Watt W. Webb).
The implant has allowed researchers to image the spinal cord for more than five weeks without affecting motor function or causing nerve damage. Rods in the chamber allow researchers to suspend the mouse above the operating table, so breathing and other movements do not interfere with the imaging.
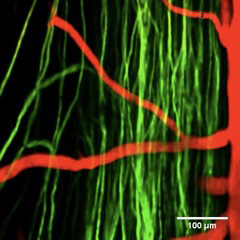
"With this procedure, we can study the same animal and look at whether axons (nerve cells) are dying back [gradual dying, beginning at the tips] or growing over time," said Chris Schaffer, associate professor of biomedical engineering and the paper's senior author. "It opens the door to new classes of experiments."
"Our method provides a platform for rapid evaluation of the efficacy of different therapeutic strategies," said Matthew Farrar, a graduate student in Schaffer's lab and the study's lead author. Joseph Fetcho, a professor of neurobiology and behavior, is also a co-author.
The new method allows continuous observations of axon behavior. Previously, researchers had to conduct repeated surgeries to check the status of axons, which provided only a snapshot of the healing process and made it difficult to determine if axons were growing or dying back.
Preliminary data confirm previous observations that the inflammatory cells of the spinal cord (microglia) surge in number immediately after trauma, while axons show varied behavior -- some begin to die back while others soon start to regrow, but the growth tapers off after eight days. Such insights may help researchers determine when and where to focus efforts when it comes to regrowing severed nerve cells.
"Axons that manage to persist near the injury may have the best chance of regrowing," said Schaffer. "Growing back every axon is probably not necessary for some recovery of function, and getting back just a few may be valuable."
Also, the new procedure offers insights into the optimal time for treating a spinal injury. The priority for clinicians has been to stabilize a patient immediately following spinal trauma, but they may want to also apply therapies early in the recovery process, Farrar added.
Without continuous long-term observations, early readings also may lead one to "falsely conclude that a therapy is more successful than it really is," said Farrar.
The study was funded by the National Institutes of Health and the National Science and Research Council of Canada.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe