Engineered molecules tag proteins for destruction
By Anne Ju
Like little mercenaries following strict orders, Cornell-developed engineered molecules called “ubiquibodies” can mark specific proteins inside a cell for destruction, paving the way for new drug therapies or powerful research tools.
Chemical engineers led by Matthew DeLisa, the William L. Lewis Professor of Engineering, have developed a new type of antibody, called a “ubiquibody,” which is an antibody fragment they have inserted into the natural process known as the ubiquitin-proteasome pathway (UPP). Their work appears in the March 16 issue of the Journal of Biological Chemistry and is highlighted in an analysis piece in the publication SciBX.
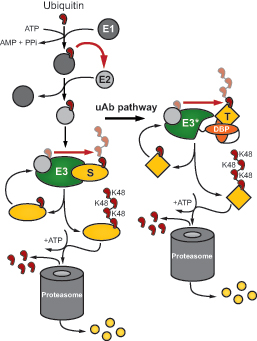
The UPP is the natural cellular pathway, or process, by which a cell gets rid of proteins it doesn’t want anymore. A doomed protein gets tagged with a chain of a protein called ubiquitin, which is like a molecular sign that reads, “destroy me.” The ubiquitin-tagged protein gets sent to the cell’s proteasome – the cell’s trash compactor – which breaks the protein into component amino acids.
DeLisa and colleagues hypothesized that this common process could be harnessed as a simple, tunable way to eliminate certain target proteins in a cell without having to mess with the genome to delete the protein using standard genetic engineering tools.
They did it by taking advantage of the modular nature of the UPP, which involves three enzymes called E1, E2 and E3. They modified a particular E3 enzyme called CHIP, giving only that part of the pathway a makeover.
They removed CHIP’s natural binding domain, replacing it with an engineered binding protein – in this case an antibody fragment – that was created in the lab. The idea was to empower CHIP to put ubiquitin chains on any target, guided by the homing capabilities of the antibody fragment to seek out and bind to its specific target. They named the entire re-engineered molecule with the modified CHIP enzyme a ubiquibody.
To prove their concept, the researchers modified CHIP with a binding protein that targets the enzyme beta-galactosidase. They introduced DNA that encoded for their beta-galactosidase target into a human cell line, along with DNA that encoded their ubiquibodies with a binding protein for the beta-galactosidase enzyme. Sure enough, beta-galactosidase levels went down in the presence of the corresponding ubiquibodies.
“Our ability to redirect whatever protein you want to the proteasome is now made possible simply by swapping out different binding proteins with specificity for targets of interest to the researcher,” DeLisa said.
Ubiquibodies could provide a powerful way to not only completely delete a protein from a cell to study that protein’s effects, but to discover what happens if, say, only 50 percent of that protein is deleted. Current gene knockout technologies are all or nothing, DeLisa said. Ubiquibodies could fine-tune research around protein deletion or reduction.
The technology could also prove useful for future drug therapies. In a cancer cell in which a certain protein has been identified as contributing to the disease, the ubiquibody could reduce or eliminate the protein from within by targeting that specific protein only, DeLisa said.
The therapeutic potential for ubiquibodies is being explored further in DeLisa’s lab, with experiments on target proteins known to be present in diseases including Alzheimer’s, cancer and Parkinson’s.
First author of the paper, “Ubiquibodies, Synthetic E3 Ubiquitin Ligases Endowed with Unnatural Substrate Specificity for Targeted Protein Silencing,” is Alyse Portnoff, a former Cornell biomedical engineering graduate student. Other co-authors are Erin Stephens, a graduate student in the field of biochemistry, molecular and cell biology; and Jeffrey Varner, professor of chemical and biomolecular engineering.
The work was supported by the National Institutes of Health.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe