Insights from patient who cleared hepatitis C could lead to vaccine
By Krishna Ramanujan, Cornell Chronicle
By studying individuals who spontaneously clear hepatitis C infections, a team of researchers has identified viable vaccine targets for a disease that infects 70 million worldwide with case numbers increasing every year.
It turns out that a quarter of people who become infected with the hepatitis C virus clear the infection on their own without treatment, while the remaining three-quarters of people develop chronic infections that can last for years. The blood-borne disease – which causes liver cirrhosis, liver failure and liver cancer – is especially prevalent among people who inject drugs.
Direct-acting antivirals developed around a decade ago are 98% effective. But even so, the number of hepatitis C cases has increased year-over-year mainly because early infections are hard to detect, access to treatment is limited and reinfections occur even after treatment.
“That’s why there is now a big interest in developing a hepatitis C vaccine,” said Andrew Flyak, assistant professor of microbiology and immunology in the College of Veterinary Medicine and co-corresponding author of the study, “Convergent Evolution and Targeting of Diverse E2 Epitopes by Human Broadly Neutralizing Antibodies are Associated with HCV Clearance,” which published March 21 in the journal Immunity. Justin Bailey, associate professor of medicine at Johns Hopkins University, is co-corresponding author.
“Our study gives us a glimpse into how certain individuals clear a highly variable infection, and we believe this information can inform a vaccine development,” Flyak said.
The study was made possible due to the unique access that Bailey had to samples from people who injected drugs and were at risk of acquiring the virus. This allowed the researchers to track individuals who were hepatitis C negative when they enrolled in the program, and to see upon subsequent clinic visits whether that person acquired the virus. Bailey obtained samples from individuals who cleared the infection on their own and those who developed chronic infection.
Viruses that evolve very rapidly, such as SARS-CoV-2, influenza and hepatitis C, have extraordinary genetic diversity with multiple strains. Combating these types of infections requires special antibodies (blood proteins that recognize pathogens and neutralize them) called broadly neutralizing antibodies (bNAbs), which can neutralize diverse viral variants.
In previous studies, researchers isolated bNAbs from people who were chronically infected with hepatitis C virus. They found that their bNAbs were using a single antibody gene to encode a variable part of the antibody molecule.
“In order to make an antibody, immune systems use multiple sets of different antibody genes, but for whatever reason the immune systems in people with chronic hepatitis C infections used just one variable antibody gene, called VH1-69,” Flyak said. Also, most of the bNAbs from these chronically infected donors targeted a specific region of the hepatitis C virus, namely the front layer of the so-called E2 protein. The immune system in chronically infected individuals has failed to clear the virus.
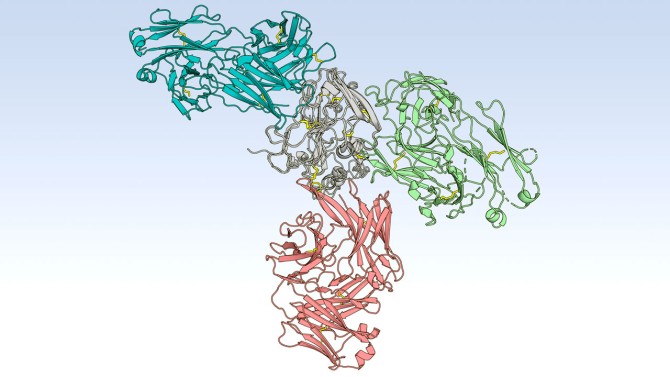
In the current study, the researchers isolated bNAbs from one person who spontaneously cleared three separate infections. This individual’s bNAbs revealed important distinctions. First, these bNAbs were genetically diverse, meaning they are encoded by a variety of variable genes, and not just one segment of the VH1-69 gene. Second, bNAbs from this individual targeted three different regions of the virus’ E2 protein, the front layer, as well as a back layer and a b-sandwich.
The data suggests that a hepatitis C virus vaccine should elicit bNAbs to all three regions of the E2 protein rather than just one region of the virus, Flyak said.
“If you have a response to multiple regions, you can have a synergistic effect, you get a response that is much stronger than the sum of its parts,” he said.
BNAbs from the individual who cleared the infections also revealed evidence of what is called convergent evolution, where different bNAbs have the same mutations but come from different antibody variable genes. “You see the same mutations in two different broadly neutralizing antibodies – it means those mutations are important,” Flyak said – and they increase the breadth of the antibody response to hepatitis C virus.
Members of Flyak’s lab used X-ray crystallography to solve the crystal structures of bNAbs in complex with hepatitis C virus’ E2 protein and show how bNAb’s mutations interact with the E2 protein. “That information can be used to design better vaccine candidates,” Flyak said.
In next steps, the team will collaborate with a larger international group to screen multiple vaccine candidates in animals and eventually identify which ones to bring into human clinical trials.
Clinton Ogega, a former graduate student in Bailey’s lab, is the paper’s first author. Two postdoctoral fellows in Flyak’s lab, Marty Schoenle and Xander Wilcox, contributed to the study.
The study was supported by the National Institutes of Health.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe