Back, neck pain sufferers could find relief with Cornell-developed spinal disc implants
By Anne Ju
Every year, millions of people see doctors for lower back and neck pain. Cornell engineers in Ithaca and doctors at Weill Cornell Medical College have created a biologically based spinal implant that could someday mean relief for countless sufferers.
Larry Bonassar, associate professor of biomedical engineering and mechanical engineering, and Roger Härtl, associate professor of neurosurgery at Weill Cornell Medical College and chief of spinal surgery at NewYork-Presbyterian Hospital/Weill Cornell Medical Center, have created an artificial implant to replace intervertebral discs in the spinal column. Their work is published online July 28 in Proceedings of the National Academy of Sciences.
In the body, the discs lie between adjacent vertebrae in the spine and form a cartilaginous joint to allow movement. The discs can degenerate or become injured, leading to severe back and neck pain due to vertebrae rubbing together, or they can herniate and cause compression of nerves or the spinal cord.
The scientists began collaborating on the project in 2006, first funded by an Ithaca-Weill seed grant. Since then, the project has moved into animal testing stages and has received several awards and accolades, a $325,000 grant from Switzerland's AOSpine foundation and $100,000 in support from NFL Charities.
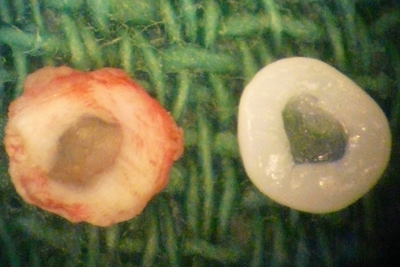
Human discs look something like a tire, with the outer part, called the annulus, made of a stiff material, and the inner circle, the nucleus, made of a gel-like substance that gets pressurized and bears weight.
Bonassar's lab, which focuses on the regeneration and analysis of musculoskeletal tissue, engineered artificial discs out of two polymers -- collagen, which wraps around the outside, and a hydrogel called alginate in the middle. They seeded the implants with cells that repopulate the structures with new tissue. Remarkably, as opposed to artificial implants today that degrade over time, the scientists are seeing that the implants get better as they mature in the body, due to the growth of the cells.
"Our implants have maintained 80-90 percent of initial disc height," Bonassar said. "In fact, the mechanical properties only get better with time."
The implants would treat a broad category of illness called degenerative disc disease -- a leading cause of disability worldwide. According to Härtl, an increasing number of patients need treatment or surgery from the degeneration of the intervertebral disc. A surgical procedure approved by the FDA in 2005 involves removing the disc completely and replacing it with an implant made of a combination of metal and plastic, with the aim of mimicking the normal movement of the lumbar and spine.
"Bone or metal or plastic implants are complicated structures that come with a mechanical risk of the structures moving around, or debris from the metal or plastic particles accumulating in the body from wear and tear," Härtl said. From a biological perspective, Härtl said, the new discs could create a "huge advantage" over traditional implants because of how they integrate and mature with the vertebrae. This major surgery, he said, would be less invasive, safer and come with fewer long-term side effects.
The scientists are hoping to move their experiments into larger animals, possibly working with Cornell's College of Veterinary Medicine, with the eventual goal of implanting the devices in humans.
Get Cornell news delivered right to your inbox.
Subscribe