Cryo-electron microscopy sheds new light on batteries
By Tom Fleischman
The interface of the solid anode and the liquid electrolyte plays a crucial role in the performance of a lithium-metal battery, but characterizing the processes that happen at that intersection has been a challenge.
To study the surface of the anode, generally the liquid electrolyte is removed and the surface is washed and dried before analysis. But this washing and drying fundamentally changes the structure and chemistry of the interface; to get an accurate picture of the interface, it must be viewed in its natural state.
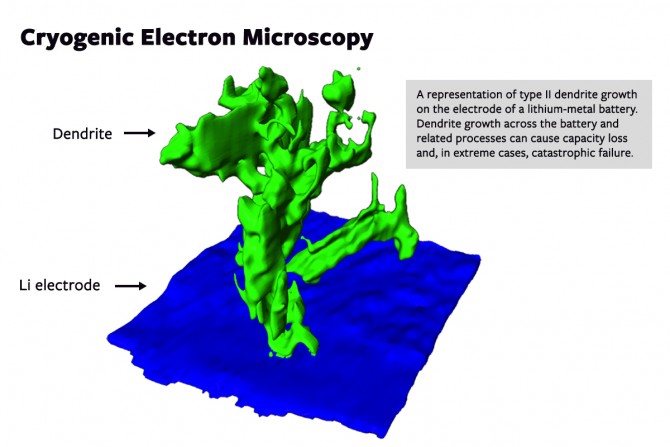
Researchers in the lab of Lena Kourkoutis, assistant professor of applied and engineering physics, have developed and demonstrated a technique for direct visualization of solid-liquid interfaces in an effort to better understand a major problem with Li-metal batteries: dendrite growth on the anode, which can cause short-circuiting and, in extreme cases, catastrophic battery failure.
Michael Zachman, Ph.D. ’18, a member of the Kourkoutis lab, is lead author of “Cryo-STEM Mapping of Solid-Liquid Interfaces and Dendrites in Li-Metal Batteries,” which will publish Aug. 15 in Nature.
Key contributions were made by the lab of Lynden Archer, the James A. Friend Family Distinguished Professor of Engineering in the Smith School of Chemical and Biomolecular Engineering. Zhengyuan Tu, Ph.D. ’17, and Snehashis Choudhury, Ph.D. ’18, both of the Archer group, constructed and analyzed the performance of the batteries used in the study.
The method developed by Kourkoutis’ lab involves rapid freezing of the electrolyte on the electrode, and a suite of cryogenic microscopy techniques to analyze the morphological, chemical and structural information at the solid-liquid interface. This work has implications for systems well beyond energy storage, the researchers say.
“The technique we developed really just allows us to have an undistorted view of what is going on at these very complex interfaces,” Kourkoutis said. “And that is key in order to understand not only that particular interface, but also the implications of the reactions or processes that occur.”
Kourkoutis said this work was inspired by her experience in a biology lab at the Max Planck Institute in Germany, where she used a method called cryo-FIB (focused ion beam) to look at processes inside cells. At Cornell, her group adapted cryo-FIB for solid-liquid interfaces and combined it with cryo-STEM (scanning transmission electron microscopy) to access the intact structure of dendrites at the nanoscale.
For this work, coin cell batteries were opened and the electrode immediately plunged into a cryogen to rapidly freeze and preserve the structure. Zachman, who prepared the samples and performed the experiments, discovered two distinct types of dendrites on the anode surface: Type I was relatively large (approximately 5 microns across) with low curvature; type II was hundreds of nanometers thick and tortuous.
In addition, the type I dendrites displayed an extended solid-electrolyte interphase (SEI) – a soft layer thought to be a precursor to dendrite growth – approximately 300 to 500 nanometers thick, much larger than has been previously observed. Discovery of this layer – which the study suggests is mostly lost during the washing and drying necessary in traditional analysis – signals that more lithium is irreversibly lost to the SEI layer than previously thought.
The group’s technique also revealed that the type II dendrites were composed of lithium hydride. “Only dendrites composed of lithium metal were assumed to be in batteries,” Zachman said, “and now we see that there are actually lithium hydride dendrites present as well, and they should have significant performance effects on the battery.”
Archer said these discoveries should help provide “important clues about how one might approach chemical design of battery electrolytes.”
The collaboration between the Kourkoutis and Archer groups stemmed from a joint proposal written to secure $2.7 million from the National Science Foundation to obtain the scanning transmission electron microscope used in this research.
“This is an outstanding demonstration of the legacy of radical collaborations that have come to define materials science research at Cornell, and which differentiate Cornell from its peers as the place to do such work,” Archer said.
This work was supported by grants from the National Science Foundation, the Packard Foundation and the Department of Energy. It made use of the Cornell Center for Materials Research Shared Facilities, an NSF Materials Research Science and Engineering Center, as well as the KAUST-CU Center for Energy and Sustainability, which is supported by a grant from the King Abdullah University of Science and Technology. Additional support came from the Kavli Institute at Cornell for Nanoscale Science and the Energy Materials Center at Cornell.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe