Mutant proteins linked to DNA damage, muscular dystrophy
By David Nutt
New Cornell-led research has found a strong connection between DNA damage triggered by mutations in proteins that surround the cell nucleus, known as lamins, and muscular dystrophy. By exploring the mechanisms that cause the damage, the researchers are hopeful their discovery can help shape better treatments.
The team’s paper, “Mutant Lamins Cause Nuclear Envelope Rupture and DNA Damage in Skeletal Muscle Cells,” was published Dec. 16 in Nature Materials.
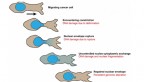
The vast majority of muscular dystrophy cases are caused by mutations in dystrophin proteins, which sit at the surface of muscle cells and, when healthy, hold the cells together. Jan Lammerding, associate professor in the Meinig School of Biomedical Engineering, has long looked to the thin membrane, called the nuclear envelope, that surrounds the cell nucleus to understand how it, too, can cause certain forms of muscular dystrophy when components of it are mutated – specifically in muscle cells, which are sensitive to mechanical stress.
“There are more than 500 different mutations that have been identified that cause different diseases,” said Lammerding, the paper’s senior author. “Even though these proteins are expressed in all cells in the human body, most of the mutations cause only muscle disease. So it’s always been a mystery: Why is it that only muscle cells show defects while, for example, the brain, the liver and other tissues that also have the mutant proteins do not show any defects?”
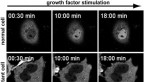
To understand how these mutated lamins can lead to degenerative muscle disease, Lammerding found inspiration in another long-term research project in his lab: tracking cancer cell migration. Three years ago, his team discovered that when cancer cells invade other tissues, the action of squeezing through tight spaces forces their nuclear envelopes to rupture, damaging the DNA. As part of that work, Lammerding’s team developed fluorescent reporter proteins to visualize the nuclear envelope rupture and DNA damage. Now they have taken those fluorescent reporters and used long-term time-lapse microscopy to systematically track the movement of nuclei in skeletal muscle cells.
“Normally one of the challenges with studying muscle diseases in vitro [outside a living organism] or in a cell culture dish is that you very rarely can get nice mature muscle fibers, similar to what you would see in vivo [inside the living organism],” said co-lead author and postdoctoral researcher Tyler Kirby. “So we worked with a group in Portugal to develop a 2.5D protocol to get highly contractile mature muscle fibers. It’s more of a tissue engineering approach, and that allowed us to be able to study the long-term effects of these mutations and how they can progressively develop during the differentiation from a progenitor cell into a mature muscle fiber.”
The team found a striking correlation between nuclear envelope rupture, DNA damage and disease severity in three mouse models. Lammerding realized the real culprit was the movement of nuclei within the muscle cells.
“We had always assumed it’s the muscle contraction that’s responsible for damaging the nuclei,” Lammerding said. “Then Tyler did a number of experiments that used drugs that could completely block the muscle contraction, and we still saw massive amounts of nuclei damage, which was very surprising to us. When we looked more closely, we recognized that the damage occurred as the nuclei move around.”
Now that the team understands the mechanism behind the DNA damage, it is exploring several therapeutic treatments. One approach involves severing the molecules that connect the nucleus to the cell’s cytoskeleton. This physical decoupling protects the nucleus from mechanical stress on the muscle fiber and prevents nuclear envelope rupture.
Another option is to suppress the signaling that occurs when the nuclear envelope ruptures and DNA becomes damaged. Often, the cell’s response to this damage is apoptosis, or self-programmed cell death. While in many other scenarios this process helps to prevent cancer, in muscle cells this damage response, more than anything, is detrimental to the cell. The researchers hope that by targeting specific molecular pathways, they can inhibit cell death in the muscle tissues without making the patient more vulnerable to cancer.
Lammerding’s team is also investigating whether the findings stretch beyond skeletal muscle and apply to heart muscles as well.
The paper’s other co-lead authors are Ashley Earle, Ph.D. ’19, and postdoctoral researcher Gregory Fedorchak, Ph.D. ‘18. Former visiting scientist Philipp Isermann, Jineet Patel ’18, Sushruta Iruvanti ’19 and researchers from the University of Iowa and the Myology Institute, Paris also contributed to the paper.
The research was supported by the National Institutes of Health, the Department of Defense Breast Cancer Research Program, the National Science Foundation, the Muscular Dystrophy Association and gifts from the Mills family. Part of the work was performed at the Cornell NanoScale Science and Technology Facility, a member of the National Nanotechnology Coordinated Infrastructure, which is supported by the National Science Foundation.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe