Philippa Johnson in her lab at the College of Veterinary Medicine.
Radiologist creates brain atlases for cats, dogs, horses
By Olivia M. Hall
A clinical radiologist in the College of Veterinary Medicine (CVM) has created three brain atlases – feline, canine and equine – to help improve MRI diagnostics.
“Currently, our state of neuroimaging is to perform standard MRIs,” said Philippa Johnson, assistant professor in CVM’s Department of Clinical Sciences. “But there are limitations, because readouts can be variable between radiologists, and some of the lesions – such as in epilepsy – aren’t visible on standard MRIs. The diagnosis is therefore presumptive.
“I wanted to start applying the most advanced forms of neuroimaging currently being done in human clinical research in the veterinary world. The big tool that we were missing was the brain atlas,” she said.
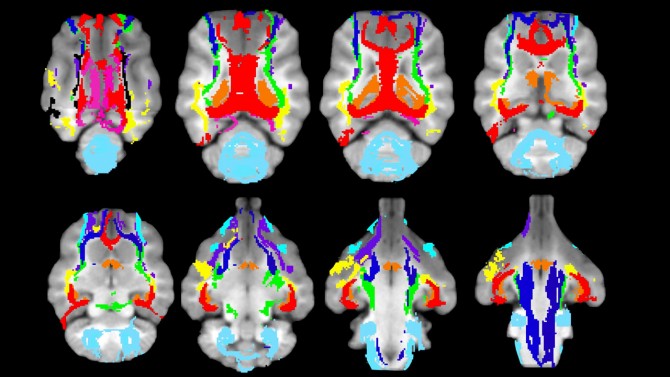
Cutting-edge, high-resolution stereotaxic brain atlases bring data obtained from multiple individuals into a standardized virtual space.
“There they can be compared to each other,” Johnson said. “The atlas also enables us to identify a standardized region in the brain that we want to compare across subjects.”
She hopes that the newest publications – freely available for download online – will enhance existing tools and fill in significant gaps.
Supported by the Harry M. Zweig Memorial Fund for Equine Research, Johnson and her colleagues used powerful 3-Tesla magnets, with twice the power of routine MRI devices, to image the brains of 15 horses euthanized for reasons unrelated to the study. The team also equipped the equine brain atlas with subcortical masks, a feature that lets users separate out specific parts of the brain for more detailed insights.
Until recently, such cortical gray matter masks were missing from all existing brain atlases for dogs, who frequently serve as models for human disease because of their long life spans and similar health issues.To fill in the gap, Johnson and her team, in collaboration with University of Sydney ophthalmologists Drs. Kathleen Graham and Andrew White, collated data from 40 dogs. They divided their new atlas into 234 regions, based on intricate drawings of the brain’s myeloarchitectonics – segmentation of the cortex according to the cellular architecture or the tissue.
“Each region is likely to have a specific function, but at this stage we don’t know yet what these are,” Johnson said. “We’re going to understand more as we do more functional imaging of the canine brain.”
The gray matter mapped so far is “where all the thinking happens,” she said. Now, the team has shifted its focus to white matter, which transports the messages to different parts of the brain or down the spinal cord. Johnson and her colleagues are mapping major white matter pathways using diffusion tensor imaging (DTI), an advanced MRI technique that detects different forms of water diffusion in tissue and can be used to virtually dissect white matter with the help of tractography, a 3D-modeling process.
DTI tractography also produced the most recent feline brain atlas, which Johnson published in mid-March. Users can compare cats’ brain scans to the atlas to pinpoint which regions show changes with certain diseases.
“We can see, for instance, which pathways are affected by feline aging,” Johnson said. “Or say I have a group of feline epilepsy cats, and all of them have these changes within their brains over this specific region of white matter. What region is that?”
While Johnson would have preferred to work with data from more than eight cats for the feline atlas – human brain templates may be created from as many as 250 individuals – feedback from colleagues around the world has been positive, she said.
“Each manuscript has its own specific purpose, and they can always be improved on,” she said. “Our lab really welcomes collaboration and data sharing, so we’re very open to different labs reaching out to us. It’s nice to be able to support the application of advanced MRI techniques in veterinary neurology, and we’d like to promote this level of research as much as possible.”
Olivia M. Hall is a freelance writer for the College of Veterinary Medicine.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe