Researchers connect matrix fiber structure and cell behavior
By David Nutt
Sometimes, good cells turn bad. But it’s not their fault: They are products of their environment.
Consider the case of myofibroblasts, connective tissue cells that assist in wound healing. These cells produce, bundle and pull together the collagen fibers of the surrounding microenvironment – a fibrous mesh known as the extracellular matrix, which restores the tissue in wound repair. But if myofibroblasts stick around and overstay their welcome, the tissue stiffens, producing more myofibroblasts that in turn cause more stiff tissue – a feedback cycle that ultimately can lead to diseases such as atherosclerosis and cancer.
Scientists have not known what exactly activates these cells – either changes in the stiffness of the microenvironment or structural changes of the matrix’s collagen fibers that may be caused by preexisting conditions like obesity or aging.
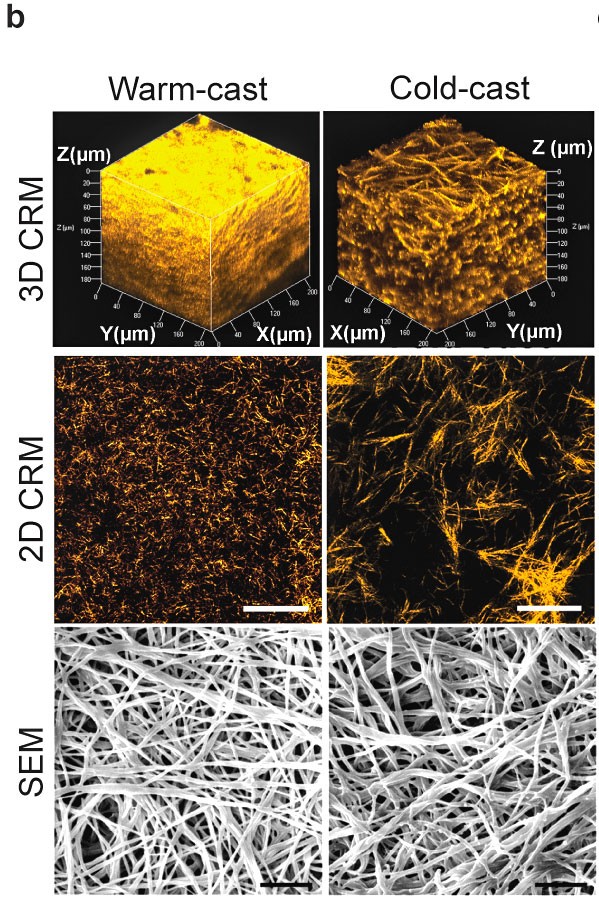
A Cornell-led collaboration investigated how differences in these collagen fibers are responsible for influencing the behavior of myofibroblasts – findings that could have implications for preventing and treating fibrotic diseases such as cancer.
Their paper, “Collagen Microarchitecture Mechanically Controls Myofibroblast Differentiation,” published May 8 in Proceedings of the National Academy of Sciences.
“For the longest time, nobody realized that stiffness, regardless of any biochemical cues, could change cell behavior. Then it became really well accepted and led to a new field termed mechanobiology,” said senior author Claudia Fischbach, professor of biomedical engineering. “However, which effect matrix structural changes alone could have on cell behavior and mechanosignaling is less understood. By integrating experimental and computational models, we were able to show how the structure of individual fibers modulates local tissue stiffness and how these changes lead to continued activation of myofibroblasts.”
Fischbach’s team modeled the mechanical interactions of cells with different matrix microarchitectures by microfabricating collagen hydrogels with varied fiber and pore size and analyzing how cells responded to these hydrogels. The team partnered with a group at the University of Pennsylvania, led by professor and co-author Vivek Shenoy, that used computational models to understand the connection between the matrix structure and the molecular machinery inside the cells, called mechanosignaling.
The researchers found that matrix networks with thicker fibers and larger pores cause cells to become more contractile. As a result, these cells pull on the fibers and align them better, which makes the tissue stiffen locally. The stiffness then activates the mechanosignaling that encourages cells to differentiate into myofibroblasts.
“One of the exciting things we’ve shown is how the cells respond to that matrix,” said Fischbach, co-director of the Cornell Center on the Physics of Cancer Metabolism. “As cells become better at pulling on fibers, they also deposit a different type of matrix subsequently. This changes the cells’ signaling, further reinforcing their response to the initial collagen structure.”
“Our results suggest if a matrix with thicker fibers is already pre-existing,” she said, “the myofibroblasts actually use it as a cue to become even more contractile and promote tumor growth and progression.”
Fischbach’s lab has a particular interest in how obesity can prime the microenvironment for aggressive cancers. Their previous research linked cancer cell growth and malignancy to thicker and aligned collagen fibers in obese fat tissue. The new findings clarify how a matrix composed of thicker fibers could lead to a vicious cycle of myofibroblast differentiation and tumorigenesis.
“Given the link between matrix microarchitecture and pathological fibrosis, matrix microarchitecture can be a prognostic marker for fibrotic diseases and a critical parameter to consider when designing matrix-based bio-constructs for cancer, stem cell and tissue engineering research,” said lead author Bo Ri Seo, M.S. ’11, Ph.D. ’14.
Co-authors include postdoctoral researcher Siyoung Choi; doctoral students Lu Ling and Adrian Shimpi; former lab members Young Hye Song, Ph.D. ’16, and Jacqueline Gonzalez ’13; Karin Wang, Ph.D. ’15, and Roberto Carlos Andresen Eguiluz, Ph.D. ’15; and researchers from the University of Pennsylvania, Leipzig University in Germany and the University of Ottawa.
The research was supported by the National Institutes of Health/National Cancer Institute; the Cornell Center on the Microenvironment and Metastasis; and the Cornell Center on the Physics of Cancer Metabolism.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe