Gut molecules may affect fattiness of the liver
By Blaine Friedlander, Cornell Chronicle
Sphingolipids – molecules ubiquitous throughout the human body, named after the Egyptian Sphinx for their complexity when scientists discovered them nearly 150 years ago – are not necessarily household conversation topics.
Despite their enigma, sphingolipids are involved in almost all aspects of human biology. They are found in all mammals but produced by select species of bacteria – and some happen to be prominent members of the gut microbiome.
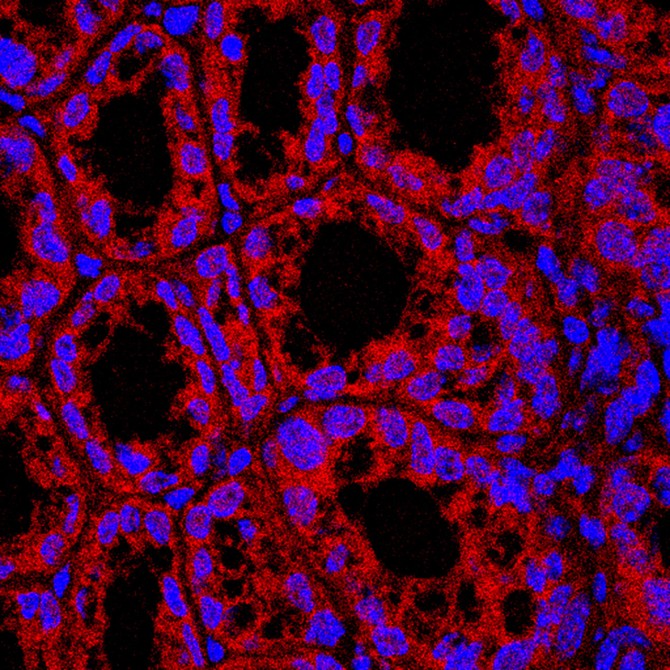
Moreover, these microbiome-derived sphingolipids appear to ameliorate a problematic fatty liver, according to Cornell research published May 26 in Cell Host and Microbes.
“Bacterial sphingolipids seem to have a role in mammalian metabolism and may help to modulate metabolic disorders,” said senior author Elizabeth L. Johnson, assistant professor of nutrition in the College of Agriculture and Life Sciences.
In June, the Pew Charitable Trusts named Johnson a 2022 Pew Scholar in the Biomedical Sciences, which provides research funding to young investigators who show promise in science relevant to the advancement of human health. She was selected from among 197 nominations submitted. Johnson will receive four years of funding for exploratory research.
Few metabolites – such as sphingolipids, with known bioactive properties – have been traced from specific microbes into host tissues. Here, the researchers labeled the sphingolipids to follow them into mouse colons and livers, where the scientists observed a protective effect on excess fat accumulation in the fattened liver – a condition known as hepatic steatosis, where triglycerides accumulate.
The sphingolipid molecules improved respiration in response to the liver’s fatty-acid overload, the paper said, suggesting that they could potentially contribute to improved, gut-mediated liver function.
“We focused on how the microbiome can produce molecules that directly interact with host tissue – such as the liver,” Johnson said. “As we figure out the process, we have a chance at controlling positive things that the microbiome is doing.”
Fatty liver disease – common in the U.S. – is a condition where excess fat builds in the human liver, according to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), of the National Institutes of Health.
Obese people (about 75%) and severely obese (about 90%) tend to get it, and up to two-thirds of type-2 diabetics get fatty livers, according to the NIDDK. About 24% of U.S. adult population has the condition.
“We were able to show that bacterial sphingolipids have a potentially wide range of function,” Johnson said, “and they move from the gut microbiome to hosts. Since gut bacteria are making this molecule, and this molecule has a function in us, then maybe this is a way the microbiome can influence human health.”
In addition to Johnson, the study, “Host Hepatic Metabolism is Modulated by Gut Microbiota-derived Sphingolipids,” was authored by Henry H. Le, postdoctoral associate, Division of Nutritional Sciences (College of Human Ecology); graduate student Min-Ting Lee and laboratory technologist Kevin Besler.
The research was supported by the National Institute of General Medical Sciences of the National Institutes of Health and by the CIFAR Azrieli Global Scholars program, Toronto, Canada. The Division of Nutritional Sciences is shared jointly between CALS and the College of Human Ecology.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe