Cornell chemists contributed to Nobel Prize-winning work
By David Nutt, Cornell Chronicle
As doctoral students nearly 20 years ago, two Cornell researchers played an early role in the development of the work that was awarded the 2022 Nobel Prize in chemistry.
This year’s honorees – Carolyn R. Bertozzi, Morten Meldal and K. Barry Sharpless – received the prize for their efforts in pioneering click chemistry and bioorthogonal chemistry. Click chemistry, developed by Sharpless and Meldal, enables molecules to snap together quickly and efficiently, despite the general messiness of most chemical reactions. Bertozzi expanded on that work by developing bioorthogonal chemistry, which applied click reactions to living organisms.
Jeremy M. Baskin, associate professor of chemistry and chemical biology in the College of Arts and Sciences and member of the Weill Institute for Cell and Molecular Biology, and Pamela Chang, associate professor of chemistry and chemical biology (A&S) and associate professor of microbiology and immunology in the College of Veterinary Medicine, were doctoral students in Bertozzi’s lab at the University of California, Berkley, in the mid-2000s. Baskin and Chang were lead authors on papers that reported some of the advances in bioorthogonal chemistry that were recognized by the Nobel announcement.
“Our goal was to develop chemical reactions for tagging molecules inside of living cells. They sort of need to click together like the parts of a seatbelt, or like two pieces of a jigsaw puzzle, where the only two pieces that are going to go next to each other are the right two, in a sea of a million other pieces,” Baskin said. “Living cells have tons of molecules in them. They have all kinds of different reactive groups. So you have to have extremely selective reagents to be able to have a labeling reaction that tags only the one molecule you want, and not kill the cell.”
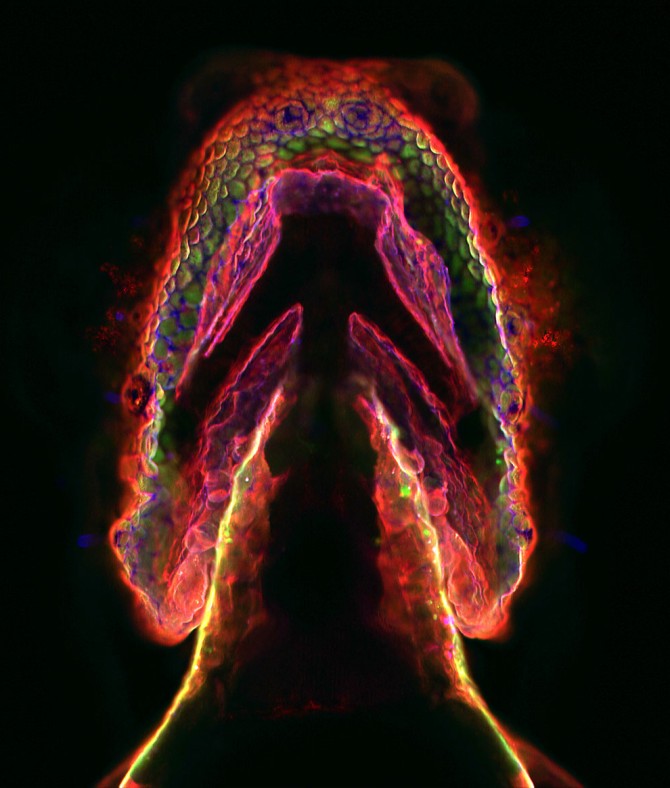
Baskin created optimized reagents for nontoxic, copper-free click chemistry tagging of glycans in zebrafish; Chang showed that the reaction works in mice. The applications of these approaches range from imaging molecules and tracking their movements in cells, to delivering diagnostic or therapeutic reagents to diseased tissue. Recent research and clinical trials are using the technique to selectively target and destroy tumors.
When Chang learned that Bertozzi had won the Nobel, she was thrilled, but it didn’t come as a total surprise. By the time she completed her Ph.D. in 2010, she was already seeing other research groups in chemistry and even other fields adopting their chemical reactions, which are now prevalent in industry as well.
“There have been some murmurings of [the prize],” Chang said.
Chang and Baskin praised Bertozzi, who’s now a professor at Stanford University, as a supportive mentor who motivated and inspired them, and also gave them the freedom and encouragement to pursue their own interests.
“She still mentors us, even as we are faculty members ourselves,” Chang said.
Chang’s experience in Bertozzi’s lab inspired her to focus on biology – specifically immunology – in her postdoctoral studies, and it continues to inform her research today.
“Two areas in my lab combine my expertise in chemistry that I developed in Carolyn’s lab and what I learned in my postdoc,” she said. “We’re essentially using click chemistry to look at enzymatic activities within the gut microbiome, which really wasn’t possible before the development of click chemistry. Everybody in my lab is a click chemist.”
Baskin recently discovered another Cornell connection to click chemistry and bioorthogonal chemistry – one that was buried in a sidenote.
“These reactions are all basically reimaginings and reinventions of very old, synthetic organic chemistry reactions that were published many decades before, but they were never tried in biological settings,” he said. “And it turns out that the reaction that I worked on in my Ph.D. and was highlighted as part of the Nobel announcement – one of the first instances of it was actually carried out in the ’50s in the Cornell chemistry department.”
The 1953 paper Baskin found was published by Alfred Blomquist, professor of chemistry, and Liang Huang Liu, a female student who went on to have an illustrious career as a chemist in China. The paper reports the first reaction of cyclooctyne with phenyl azide – the same organic chemistry reaction Baskin developed into copper-free click chemistry in the early 2000s.
“You’re always standing on the shoulders of giants, right?” he said.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe