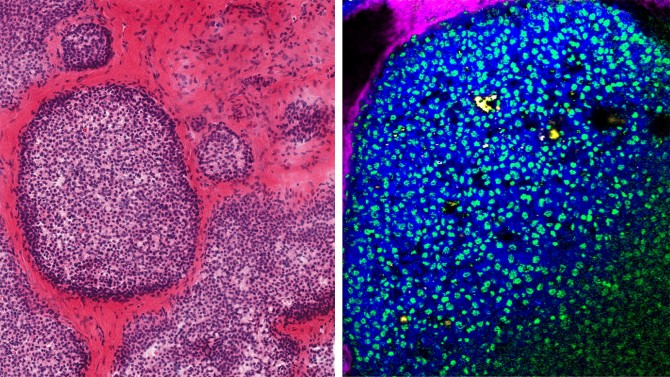
An interdisciplinary project is looking at calcium mineral deposits (called microcalcifications) associated with breast cancer and using materials science approaches to “fingerprint” the calcifications to look for clues about their pathology.
Microcalcification ‘fingerprints’ can yield info about cancer
By David Nutt, Cornell Chronicle
An interdisciplinary collaboration 10 years in the making used a materials science approach to “fingerprint” the calcium mineral deposits known as microcalcifications that reveal pathological clues to the progression of breast cancer and potentially other diseases.
The group’s paper, “Biomineralogical Signatures of Breast Microcalcifications,” published Feb. 22 in Science Advances. The lead author is postdoctoral researcher Jennie Kunitake, Ph.D. ’21.
Healthy mineral deposition is a delicately orchestrated process, as seen in bone and tooth formation. Sometimes, however, mineral deposits form in places they don’t belong, such as kidneys – i.e., kidney stones – and breast tissue. In the context of breast cancer, microcalcifications are a critical screening tool because they appear as vivid white specks in mammograms and, in certain cases, indicate the presence of breast cancer.
“Usually after the initial mammogram, microcalcifications are largely ignored. And what we’re saying is we can look beyond the resolution of the mammogram, at the microscopic and chemical level, and get more information from these microcalcifications,” said co-senior author Lara Estroff, professor of materials science and engineering in Cornell Engineering. “By taking well-established, high-resolution characterization techniques from materials science and coupling those with an appreciation for biomineralization and how organisms can control the deposition of mineral, we’ve gained a unique insight into a pathological mineral that may have important implications for disease.”
Estroff’s group specializes in biomineralization, i.e., how biological organisms control the growth of crystals in their tissues. More than a decade ago, she began collaborating with Claudia Fischbach, the Stanley Bryer 1946 Professor of Biomedical Engineering and a co-senior author of the paper, to explore the metastatic spread of breast cancer to bone. This led to an exploration of a “bizarre” phenomenon in which bone-like mineral appeared at primary tumor sites, and from there the collaborators became interested in the ways these microcalcifications can capture elements of the tissue microenvironment where they form, almost like a snapshot. The microenvironment, also known as the organic matrix, can in turn influence the mineral’s composition, morphologies and mechanical properties.
“Minerals have different rules than biology,” Kunitake said. “Minerals forming in breast cancer could be trapping chemical information that reflect their formation environment, and that could potentially have clinical value and relevance.”
While some cancer biologists have studied microcalcifications, the phenomenon has not been explored by materials scientists.
“Biomineralization is a rather niche area that involves contributions from materials science, geology, biology and more. It’s very multidisciplinary,” Estroff said. “There’s absolutely no reason that oncologists would pay attention to the materials properties of these tiny little crystals that are appearing. I think it really took someone who had an idea of what the mineral could be offering to do this. We said, can we take everything that we know from studying physiological biominerals, and apply it now to these pathological minerals?”
Fischbach connected Estroff and Kunitake with researchers at Memorial Sloan Kettering Cancer Center, who provided tissue samples containing microcalcifications from 40 breast cancer patients.
Kunitake then began the arduous, years-long process of trying to understand exactly what they were looking at. She turned to Dr. Daniel Sudilovsky, then at Cayuga Medical Center, who helped characterize the pathology of each type of microcalcification they found.
Next, rather than grind up and homogenize the tissue samples, as other studies had done, the researchers sought to obtain high-resolution, three-dimensional maps of the chemistry of the mineral and the organic matrix, such that they wouldn’t alter the tissue structure. So they collaborated with Admir Masic of Massachusetts Institute of Technology, a co-senior author of the paper, who used a vibrational spectroscopy technique called Raman microscopy that can detect the distinct vibrational signatures of a biological molecule’s organic and inorganic chemistries, and also map where those signatures are occurring.
Kunitake then set about integrating and parsing all the data using techniques that were inspired by omics research in biochemistry and genetics.
“One way to look at data, when you have a lot of it, is to use strategies from the omics communities,” Kunitake said. “It doesn’t have to be quantitative, just to visualize how the data behaves. Using hierarchical clustering, we could look at our data as a heatmap, and that gave us an idea of how different parameters that we measured were related to one another, and how the different calcifications grouped based on their fingerprints.”
Among the researchers’ key findings: cancer-associated microcalcifications cluster into physiologically relevant groups that reflect the tissue type and local malignancy; mineral carbonate exhibits substantial variety inside the tumor; trace metals – including zinc, iron and aluminum – are enhanced in malignant-localized calcifications; and the ratio of lipids to proteins within microcalcifications is lower in patients with poor prognosis.
While the researchers are not sure if the microcalcifications form before the cancer develops or because of it, the findings indicate there is a correlation with disease severity. The researchers are hopeful the findings may also illuminate calcifications in other types of cancer, such as thyroid and ovarian cancer.
The team now plans to study a larger spread of disease characteristics, and also apply their approach to other pathological mineralization diseases, such as calcific aortic valve disease, in which mineral forms in the heart valve, or as Estroff says, “the mineral is the disease.”
Co-authors include: Lynn Johnson, director and statistical consultant of the Cornell Statistical Consulting Unit; postdoctoral researcher Siyoung Choi; Dr. Daniel Sudilovsky of Kingman Regional Medical Center in Kingman, Arizona; Dr. Neil Iyengar, a medical oncologist in the Breast Medicine Service at Memorial Sloan Kettering Cancer Center and associate professor of medicine at Weill Cornell Medicine; and researchers from Memorial Sloan Kettering Cancer Center and MIT.
The research was supported by the Human Frontier Science Program and by the National Cancer Institute’s Center on the Physics of Cancer Metabolism.
The researchers made use of the Cornell Center for Materials Research, which is supported by the National Science Foundation’s MRSEC program, and the College of Veterinary Medicine’s Animal Health Diagnostic Center.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe