Protein finding could inform new type 2 diabetes treatment
By Heather Lindsey
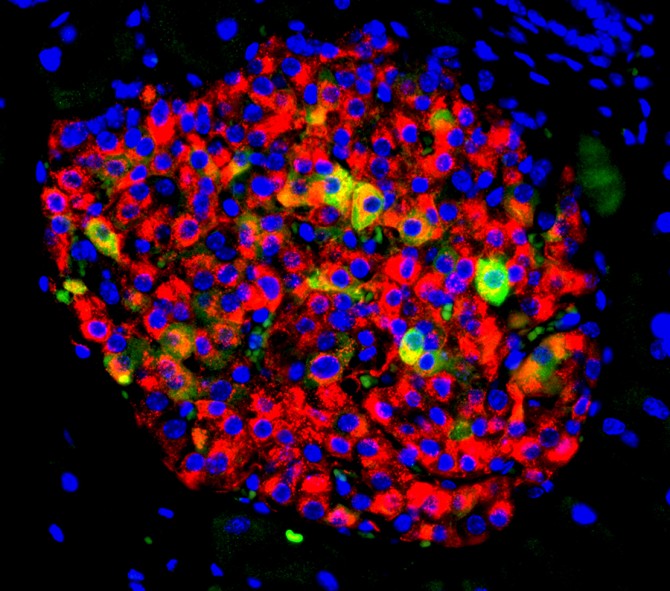
The protein adipsin helps protect insulin-secreting cells called pancreatic beta cells from destruction in type 2 diabetes, according to a new study by researchers at Weill Cornell Medicine and NewYork-Presbyterian.
The study, published Nov. 7 in Nature Medicine, may have implications for the future development of type 2 diabetes therapies that target and protect beta cells.
Adipsin is produced in body fat. Among middle-aged adults, higher levels of the protein in the blood were also associated with protection from type 2 diabetes.
“A big problem associated with type 2 diabetes is that beta cells stop functioning properly and fade away,” said senior author Dr. James C. Lo, assistant professor of medicine and of pharmacology at Weill Cornell Medicine and a cardiologist at NewYork-Presbyterian/Weill Cornell Medical Center.
About 30 million people in the United States have diabetes, and up to 95% of these individuals have the type 2 form of the disease, in which the body stops responding to insulin and pancreatic beta cells slowly stop producing enough of it.
Some of the currently available medications that target beta cells have side effects, such as lowering blood glucose levels too much, Lo said. In addition, there are no proven treatments to prevent beta cell loss. People with type 2 diabetes whose beta cells don’t work properly have to inject insulin to keep their blood glucose levels stable.
The team – which included researchers in the laboratories of Mingming Hao and Noah Dephoure, assistant professors of biochemistry, and Lukas Dow, assistant professor of biochemistry in medicine, at Weill Cornell Medicine – knew that adipsin had a role in stimulating beta cells to secrete insulin and theorized that the protein might be a potential therapy for type 2 diabetes.
To explore this theory, the scientists first conducted a study in which they increased adipsin levels in mice with type 2 diabetes. They found that adipsin had a long-term positive effect on diabetes, improving blood sugar and increasing insulin levels while helping to prevent beta cell death. “Our findings in mice showed that more adipsin in the blood translated to better diabetes control,” Lo said.
Lo and his collaborators at Icahn School of Medicine at Mount Sinai also studied human beta cells in their laboratories and determined that adipsin activates a molecule called C3a, which protects and supports beta cell function. They further discovered that C3a suppresses an enzyme called Dusp26 that can damage beta cells and cause them to die.
The researchers then directly blocked DUSP26 activity in human beta cells and found that this treatment protected the beta cells from death. Similarly, when they suppressed DUSP26 activity in mice, beta cells became healthier, meaning they could better secrete insulin.
“I hope that both adipsin or DUSP26-directed therapies may prevent type 2 diabetes patients from developing beta cell failure and from requiring insulin injections for their treatment,” said lead author Dr. Nicolás Gómez-Banoy, a postdoctoral researcher in Lo’s laboratory.
To gain a better understanding of how adipsin might be influencing the health of people in the community, the team collaborated with researchers at Harvard Medical School and Massachusetts General Hospital to evaluate 5,570 individuals enrolled in the Framingham Heart Study, an ongoing cardiovascular study being conducted in Massachusetts.
The scientists found that people with higher levels of adipsin in the blood had a lower incidence of developing diabetes in the future than people with lower levels. People with the highest level of adipsin had more than a 50% reduction in diabetes incidence compared with people with the lowest level of adipsin.
In addition, adipsin levels correlated with the amount of subcutaneous fat, which is stored just under the skin, rather than visceral fat, stored within the abdomen.
“Most people think that fat is associated with something bad, but it’s more complicated than that,” said Lo, also a member of the Weill Center for Metabolic Health and the Cardiovascular Research Institute at Weill Cornell Medicine. “Subcutaneous fat is more benign or even protective compared to visceral fat.”
Further studies are needed to determine whether a higher level of adipsin in humans protects them from developing diabetes and whether increasing adipsin levels would reduce the risk of developing diabetes in certain populations.
Lo and his research team are currently investigating whether targeting and inhibiting DUSP26 production in beta cells may be a possible avenue for drug development. “We hope this could be a novel treatment opportunity,” Lo said.
Lo and Gómez-Banoy are named inventors on a provisional patent application filed by Cornell University that covers the use of DUSP26 inhibitors for the treatment of type 2 diabetes.
Heather Lindsey is a freelance writer for Weill Cornell Medicine.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe