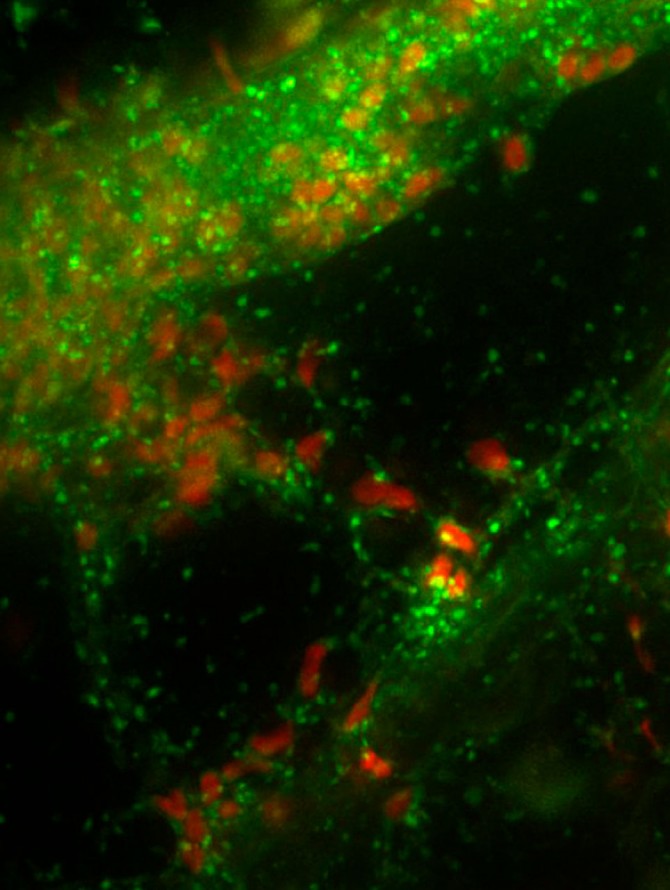
Spores and hyphae of a Glomeromycotina fungus surrounding a plant root.
Study of fungi may improve baby food, agriculture, food safety
By Krishna Ramanujan
A new grant will investigate how bacteria that live inside the cells of fungi may shape the biology, evolution, biodiversity and function of these fungi – research with important practical applications for industry, sustainable agriculture and preventing food spoilage.
Researchers from Cornell, Oregon State University and the North-West University–Potchefstroom in South Africa will team up on the four-year Dimensions of Biodiversity grant from the National Science Foundation (NSF). The funding program requires a collaborative partner from a participating country.
The two U.S. universities will share $2 million from the NSF, while North-West University researchers will receive funding from South Africa’s National Research Foundation.
“We hypothesize that these fungi are not evolving and functioning in isolation, but that they are driven by their bacterial symbionts and this is what we want to test,” said project principal investigator (PI) Teresa Pawlowska, associate professor of plant pathology and plant-microbe biology in the School of Integrative Plant Science in the College of Agriculture and Life Sciences.
Co-PIs are Jessie Uehling, assistant professor of botany and plant pathology at Oregon State University, and Rasheed Adeleke, associate professor of microbiology and soil science at North-West University.
The project will focus on three groups of fungi in the Mucoromycota lineage and bacteria that live inside their cells, called endosymbionts:
- Glomeromycotina: Also called arbuscular mycorrhizal fungi, these fungi absorb mineral nutrients, including phosphorus, from the soil and make them accessible to plant roots. Better understanding of these fungi and their endosymbiotic bacteria may offer organic farmers a natural substitute for mineral fertilizers, including phosphorus, which is being rapidly depleted.
- Mucoromycotina: Also called sugar or bread molds, these fungi are common, disperse readily via spores and cause food spoilage. They also cause mucormycosis, a rare but devastating disease that affects immunocompromised individuals and people with untreated open wounds. The infections are resistant to antifungal treatments and are lethal for half of those inflicted, but the researchers hope that better understanding of the bacterial-fungal interactions will open doors for mucormycosis treatment, as well as for limiting food spoilage.
- Mortierellomycotina: These fungi produce polyunsaturated fatty acids that are essential for brain and retina development in humans. They are also used by industries in baby formula and food supplements.
In the course of the project, the researchers will sample fungi from comparable Mediterranean-type biomes and desert biomes across the U.S. and South Africa. They will then bring samples into the lab and use DNA to identify Mucoromycota fungi and their bacterial symbionts. This information will help to investigate each bacterium’s role in evolutionary diversification and Mucoromycota community structuring.
In greenhouses, the researchers will conduct experiments to answer questions about community ecology and whether the bacteria influence fungal ability to compete for resources and space. The team will also address how genetic underpinnings and environmental factors play roles in these relationships.
“This is a novel and highly underappreciated area,” Pawlowska said, “because we did not know about these endosymbiotic relationships until recently, so it’s turning a new leaf in exploring fungal biology.”
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe